The Pharma Research Problem
Pharmaceutical research has uniquely high stakes for self-report failure.
Pharmaceutical research has relied on focus groups, patient panels, and moderated interviews for decades. The problem is structural: every one of these methods asks participants to describe their emotional response after it has occurred, in a social context, with a researcher present. The result is professionally composed, socially acceptable, and systematically distorted.
In most research contexts, self-report bias is an irritant. In pharma, it is a material risk. When patients, caregivers and HCPs cannot accurately describe their emotional responses — to a diagnosis, a treatment option, a piece of drug messaging — the consequences reach from product development to regulatory submission to commercial launch.
Consider the moments where emotional truth is critical and articulation consistently fails:
Patients in shock cannot accurately describe their emotional experience in a research debrief. The articulation gap is widest at the highest emotional intensity.
Fear, hope, confusion and resignation are present simultaneously — and none of them survive intact through a moderated discussion or a rating scale.
Healthcare professionals are trained to manage their emotional presentation. Traditional research captures the professional persona, not the genuine response to your product or messaging.
Patients say they understand treatment instructions. The emotional markers of confusion, overwhelm and doubt are present simultaneously — visible in the face, invisible in the self-report.
Pharma Use Cases
Every stage of the drug lifecycle. Every type of participant.
Patient journey interviews
Capture emotional response at every stage of the patient journey — from symptom onset through diagnosis, treatment selection, adherence and outcome. Identify the moments of highest emotional charge and lowest articulation capacity.
- Diagnosis communication emotional response
- Treatment option presentation and decision
- Adherence barrier identification
- Quality of life and caregiver burden assessment
Healthcare professional engagement
Penetrate the professional mask. EchoDepth captures the involuntary response of physicians, pharmacists, nurses and specialist prescribers to product positioning, clinical data presentation and brand messaging.
- Clinical data comprehension and trust
- Brand and product positioning response
- Prescribing decision driver analysis
- Sales aid and detail aid effectiveness
Drug messaging & communication testing
Test every variant of your drug communication — indication messaging, dosing instructions, side effect disclosure, benefit claims, patient support materials — and get moment-to-moment emotional response curves for each element.
- Benefit claim emotional credibility
- Side effect disclosure response (fear vs acceptance)
- Patient information leaflet comprehension
- DTC advertising emotional impact
Packaging & device design testing
Pharmaceutical packaging communicates trust, safety, efficacy and ease of use. EchoDepth captures the emotional response to every design element — colour, typography, hierarchy, device form factor — at the moment of first exposure.
- Pack design trust and safety signalling
- Device usability emotional response
- Self-injection confidence building
- Primary vs secondary packaging optimisation
Clinical trial participant experience
Trial participant dropout is one of the most expensive problems in drug development. EchoDepth identifies the emotional signals that predict dropout — confusion, anxiety, loss of trust — early enough to intervene.
- Informed consent comprehension and anxiety
- Protocol burden and compliance signals
- Dropout risk early identification
- Participant satisfaction and advocacy
Marketing recommendation & launch optimisation
From clinical data to commercial story to marketing recommendation. EchoDepth Insight provides the emotional evidence base for launch decisions — which messages land, which concepts resonate, which creative drives intent.
- Brand story emotional resonance testing
- Campaign concept and creative evaluation
- Launch sequencing emotional optimisation
- Post-launch emotional tracking against baseline
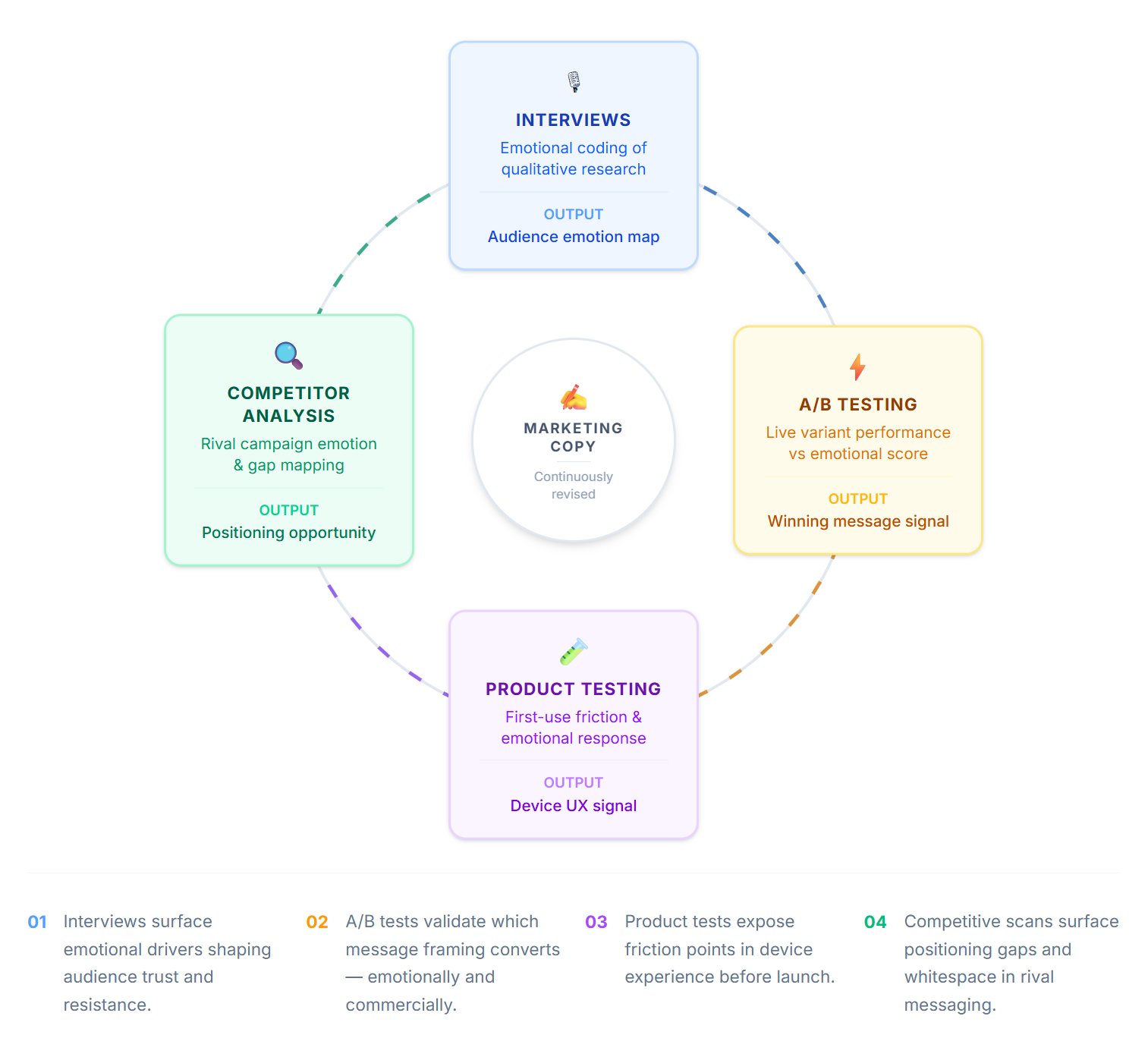
End-to-End Workflow
From emotional capture to marketing recommendation.
EchoDepth Insight is designed for the full pharma research workflow — not just the data capture moment, but the entire journey from brief to boardroom recommendation.
Research design & stimulus preparation
Work with your existing research agency or Cavefish's insights team. Define the emotional questions your research needs to answer. Prepare stimuli — concepts, messaging, packaging, video — for in-session delivery.
Remote participant sessions
Participants — patients, caregivers, HCPs — join remotely via any camera-equipped device. Consent captured. Session delivered. Emotional data captured continuously throughout, including during unstructured interview conversation.
Emotional analysis & session reporting
Every session generates a structured report: VAD scores per stimulus, emotion timeline curves, key moment flags, AU activation summaries and group aggregate analysis. Segment comparison across patient types, geographies or HCP specialisms.
Marketing recommendation & evidence base
Emotional data integrated with your qualitative and quantitative findings. A clear, evidenced marketing recommendation — which message, which concept, which creative — backed by physiological signal, not stated preference. Designed for the brand team, the commercial committee and the medical affairs review.
Common Questions
Pharma research with EchoDepth Insight.
Can EchoDepth Insight be used for patient experience or patient journey research?
Yes. EchoDepth Insight is particularly valuable for patient journey research because it captures the emotional response at diagnosis communication, treatment decision points, therapy adherence interviews and care experience evaluation — all moments where patients are unlikely to accurately articulate their true feelings.
Is EchoDepth Insight suitable for HCP research?
Yes. HCPs are a notoriously difficult research audience — their professional training makes them highly effective at managing their self-presentation. EchoDepth Insight captures the involuntary physiological signals that reveal genuine response to product messaging, clinical data presentation and brand positioning.
How does EchoDepth handle sensitive pharma research data?
No raw video is retained. Only VAD scores and Action Unit activations are output and stored. All sessions require explicit participant consent. On-premise deployment is available for organisations with strict data residency requirements. GDPR-compliant by design.
Also available
FMCG & Consumer Goods Research
The same emotional AI methodology applied to packaging concept testing, brand repositioning, advertising pre-testing and NPD research. Captures genuine purchase signal — not the "interesting but wouldn't buy" gap.
Pharma research insights
Insight
Social desirability bias in pharma research
Why patients say what researchers want to hear — and how FACS-based emotional AI detects the divergence.
Read insight →Insight
How to validate a product concept before launch
Traditional concept testing measures liking, not wanting. How emotional AI captures genuine purchase signal.
Read insight →Insight
Why focus groups fail to predict market success
Three structural flaws no facilitator can fix — and what to use instead.
Read insight →Pharmaceutical Research
Your next insight study. With emotional truth included.
Whether you're preparing for launch, optimising messaging or building your patient understanding, we can integrate emotional capture into your existing research design.